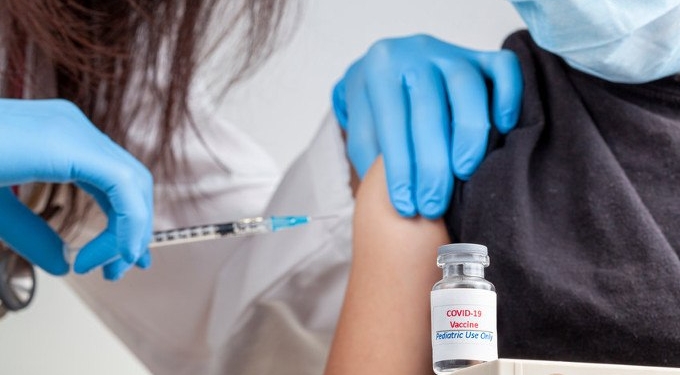
The Centers for Disease Control and Prevention’s (CDC) vaccine advisory panel today unanimously recommended Pfizer’s COVID vaccine for children 5 to 11, despite concerns raised during the meeting about Pfizer’s clinical data, the fact that children who previously acquired natural immunity to COVID were included in clinical trials and evidence showing COVID poses little risk to children.
If Dr. Rochelle Walensky, the CDC’s director signs off on the decision, children ages 5 through 11 could start receiving COVID vaccines as early as tonight.
The younger age group will receive one-third of the dose authorized for those 12 and older in two shots at least three weeks apart. The doses will be delivered by smaller needles and stored in smaller vials to avoid a mix-up with adult doses.
The CDC’s guidelines for the vaccine’s use are not legally binding, but heavily influence the medical community’s practice.
Prior to today’s decision by the CDC’s Advisory Committee on Immunization Practices (ACIP), the Biden administration enlisted more than 20,000 pediatricians, family doctors, and pharmacies to administer the vaccines — with 15 million doses already packed with dry ice, loaded into small specialized containers and shipped via airplanes and trucks to vaccination sites across the country, federal officials said on Monday.
Walensky sent a clear signal during the ACIP meeting about where she stands, CNN reported. “We have been asking when we will be able to expand this protection to our younger children,” Walensky said in opening comments to the committee.
“As you review the data today, it will be key to keep in mind the specific risks to children from this virus and the pandemic, and to put that risk into the context of other vaccine-preventable diseases,” Walensky said.
Walensky noted that children are routinely vaccinated against diseases like chickenpox — which results in far fewer hospitalizations and deaths in children compared to COVID.
During today’s meeting, ACIP members reviewed and discussed the science behind the U.S. Food and Drug Administration’s (FDA) authorization last week of Pfizer’s COVID vaccine in all children 5-11 years old.
That authorization was based mostly on a Pfizer-BioNTech study of 4,600 children worldwide, of whom approximately 3,100 got the low-dose vaccine and about 1,500 got a placebo.
These studies showed the vaccine is about 91% effective against COVID. The immune system response to the vaccine, as measured by antibodies, was comparable to the response seen in 16- to 25-year-olds, NPR reported.
During the meeting, the CDC said 745 children under 18 have died of COVID since the beginning of the pandemic — although the COVID-19 team admitted 79% were confirmed to be hospitalized for COVID, while the rest were hospital admissions for other causes.
“The chance that a child will have severe COVID, require hospitalization or develop a long-term complication like MIS-C [multisystem inflammatory syndrome] remains low, but still the risk is too high and too devastating to our children, and far higher than for many other diseases for which we vaccinate children,” Walensky said.
Efficacy of Pfizer’s COVID vaccine in children
The CDC said Pfizer’s COVID vaccine was 90.9% effective against symptomatic COVID and none of the adverse events experienced during clinical trials were assessed by “the investigator” as related to the vaccine.
To determine the efficacy of the Pfizer-BioNTech COVID vaccine, Pfizer measured the blood of 264 children for antibodies.
“There were 3,000 vaccinated children in the trial. Why isn’t blood from the other 2,700-plus being measured for antibodies?” asked Dr. Meryl Nass, a member of the Children’s Health Defense Scientific Advisory Panel.
“Pfizer never explains why, when they have an important clinical trial in which over 3,000 children were injected in this age group, only a subset of less than 10% were used to assess efficacy,” Nass said.
Nass explained:
“Pfizer claims three cases of COVID in the vaccinated group versus 16 in the placebo group show efficacy of the vaccine. But the FDA did not accept this claim. Note that all cases were mild, none hospitalized or died. So are they planning to vaccinate 28 million kids to prevent colds?”
Nass noted Pfizer also enrolled kids who had prior evidence of having had COVID in the clinical trial, “which should never have been allowed.”
“Of the kids who were already immune at the start of the trials, none developed COVID,” Nass said. “About 150 kids in the placebo group were recovered and none got COVID.”
Nass said kids with preceding COVID infection did not have their antibody levels checked after the first dose, as Pfizer stated they did not collect the data because they “tried to minimize blood draws in children.”
“The real reason they did not want to collect data is that it might support the fact that kids who already had COVID might only need one vaccine dose, or none at all,” Nass said.
During the brief public comment session, Patricia Neuenschwander, a registered nurse noted there was no prevention of hospitalization, death, or multisystem inflammatory syndrome in children — a condition being used to justify vaccinating younger children against COVID, despite numerous cases of MIS-C having been reported after receipt of a COVID vaccine.
Neuenschwander reminded the ACIP that vaccinations do not prevent infection or transmission. It is a mild illness in the vast majority of children, she said, and prior immunity is being ignored — the expansion group was only followed for 17 days.
David Wiseman, a research scientist with a background in pharmacy, pharmacology, and experimental pathology, asked the CDC panel why the efficacy study was not validated by the FDA, and why Pfizer changed the buffer [see page 14] in the vaccine but did not test it in animals or kids — planning to use an untested version of the vaccine in 5 to 11-year-olds.
Wiseman said the FDA abandoned its responsibility, and he asked if the ACIP would do the same.
Myocarditis and COVID in 5- to 11-year-olds
One side effect that generated considerable discussion at today’s meeting was myocarditis — a form of heart inflammation.
The CDC said 1,640 cases of myocarditis have been reported to the CDC’s Vaccine Adverse Event Reporting System in people under age 30 after having received a COVID vaccine, but only 877 met the CDC’s case definition.
The CDC said there were nine reported deaths in people with myocarditis, but then the agency reduced the number to three, with two cases pending evaluation and one case without adequate information.
“I have to say that it is beyond belief that CDC could whittle down 877 cases reported in young people to three actual cases. Where did the rest go?” Nass asked.
“According to the CDC’s Vaccine Safety Datalink, 7 of 16 12- to 17-year-olds with myocarditis were still on exercise restriction three months after diagnosis — that is 44% could not exercise three months later,” Nass said. “This is huge.”
Nass further noted 25% of 250 myocarditis cases were still symptomatic at three months, and only 74% of cases were designated by cardiologists as definitely resolved at 3 months.
As it pertains to safety, some who testified during a public comment period, as well as other commentators, questioned whether the study used by the FDA to grant Emergency Use Authorization is large enough to assure parents that the vaccine is safe in young children.
“The bottom line is getting COVID, I think, is much riskier to the heart than getting this vaccine,” said Dr. Matthew Oster, a pediatric cardiologist at Children’s Hospital of Atlanta.
Dr. Tom Shimabukuro covered vaccine safety monitoring from the CDC’s surveillance system in children. Shimabukuro said COVID is getting the “most intensive vaccine monitoring program in history,” yet he did not go into detail on surveillance data.
Acknowledging that some parents are hesitant about vaccinating their children right away, Dr. Matthew Daley, a member of the ACIP said, “we hear you loud and clear and of course you only want what’s best for your child. I encourage you to talk to your family physician or pediatrician, they can walk through this with you.”